Scientific analysis confirms that tetrahydrocannabinol (THC) directly manipulates the body's endocannabinoid system, triggering a phenomenon known as cannabis-induced hyperphagia. A recent 2025 cross-species study demonstrates that this neurological "hijack" amplifies both the biological drive to eat and the reward of consumption, fundamentally altering how the brain perceives satiety and food valuation.
Comparative Analysis of THC-Induced Feeding Behaviors
To understand the mechanics of the "munchies," researchers at Washington State University and the University of Calgary conducted tandem experiments on both humans and animal models. The following table outlines the behavioral outcomes based on THC exposure.
| Subject Group | Exposure Condition | Behavioral Outcome / Food Preference |
|---|---|---|
| Human Adults (n=82) | Placebo Vape (0mg THC) | Baseline caloric intake; standard dietary choices. |
| Human Adults (n=82) | Active Vape (20mg or 40mg THC) | Significant intake increase; gravitation toward savory/neutral items (beef jerky, water). |
| Animal Model (Rats) | Sober + Satiated (Full) | Ceased working (lever pulling) to obtain food. |
| Animal Model (Rats) | THC Intoxicated + Satiated | Resumed working for any food, behaving as if in starvation. |
The Neurology of Cannabis-Induced Hyperphagia
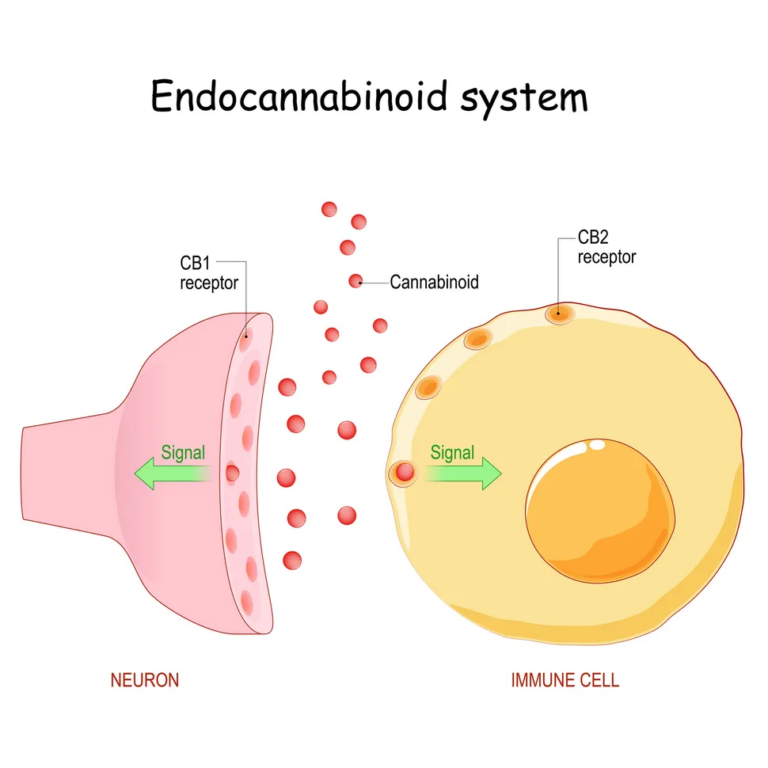
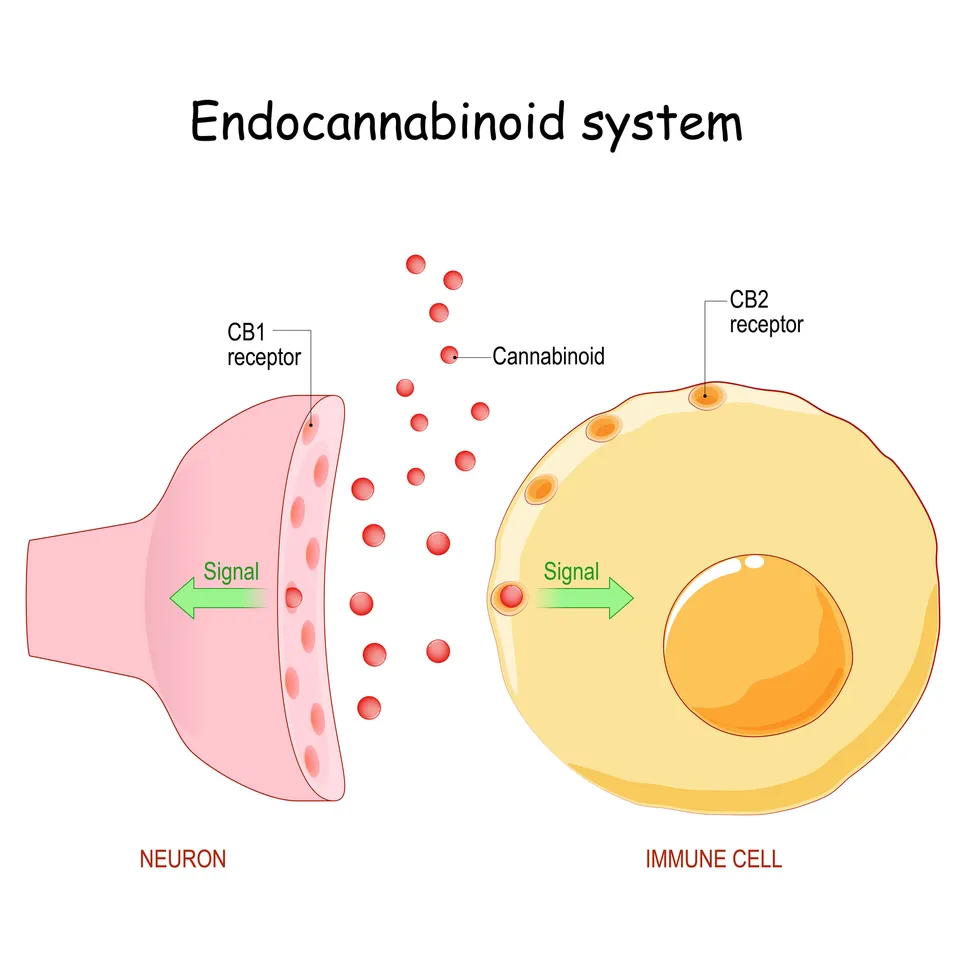
The intense increase in appetite following cannabis consumption is driven by the body's endocannabinoid system. This complex network of signaling molecules and receptors helps regulate critical functions, including mood, stress, pain, and appetite. According to Ryan McLaughlin, co-director of the Cannabis Research Center at Washington State University, endocannabinoids act like a "dimmer switch," fine-tuning neural activity to maintain biological balance, or homeostasis.
When a person consumes cannabis, tetrahydrocannabinol (THC) enters the bloodstream and mimics these natural endocannabinoids. It binds primarily to CB1 receptors in the brain, which are heavily concentrated in regions tied to craving and reward, particularly the hypothalamus—the brain's hunger coordination center.
How THC "Hijacks" Satiety Signals
Under normal conditions, the body releases endocannabinoids briefly and locally to nudge the system toward hunger. However, THC introduces a massive, systemic disruption. McLaughlin notes that THC activates these CB1 receptors "more broadly and for a longer period of time."
This prolonged activation effectively "hijacks" the appetite circuit. By artificially enhancing the motivational value of food, THC tricks the brain into believing the body is in an acute fasting state. This neurological override explains why a person can feel completely full one minute and ravenously hungry the next.
The 2025 Study: Dispelling the "Junk Food" Myth
A common cultural misconception is that the "munchies" strictly result in a binge on high-sugar, carbohydrate-rich junk foods. A 2025 study co-led by WSU and the University of Calgary challenged this assumption. In a trial involving 82 adults ages 21 to 62, participants who vaped 20mg or 40mg of cannabis ate significantly more than the placebo group—but their choices were unexpected.
Carrie Cuttler, a WSU psychology professor and study co-author, noted that beef jerky and water were among the top items intoxicated participants gravitated toward, rather than expected treats like chocolate or chips.
To isolate this variable, researchers conducted tandem experiments on rats. Matthew Hill, a professor of medicine at the University of Calgary, explained that even when rats were completely full, THC intoxication caused them to pull a lever for food "as if they're starving." Crucially, the rats did not exclusively seek out dopamine-triggering carbohydrates; they worked for any available food, proving the drive is rooted in sheer caloric intake rather than specific flavor profiles.
Variables and Alternative Appetite Triggers
While cannabis is famous for this effect, it is not the only trigger for the endocannabinoid system. Sleep deprivation, intense stress, and rigorous exercise can also activate these appetite circuits. However, THC is unique because it simultaneously enhances both the hunger-regulating regions and the reward circuits, amplifying both the biological drive to eat and the physical pleasure of eating.
McLaughlin emphasizes that the "munchies" are not a universal guarantee. The intensity of cannabis-induced hyperphagia varies wildly based on the specific THC dose, the user's tolerance, the method of ingestion, and individual metabolic makeup.
Verdict: The Future of Appetite Research
From a clinical perspective, understanding how THC manipulates the hypothalamus offers profound insights into human metabolism. Historically, data on the "munchies" relied heavily on anecdotal self-reporting. As cannabis regulations evolve, tightly controlled laboratory studies measuring actual caloric intake and hormonal shifts will replace these anecdotes. Ultimately, decoding this "hijacked" circuit could pave the way for targeted therapies for eating disorders, cachexia (wasting syndrome), and obesity, utilizing the endocannabinoid system to safely suppress or stimulate the human appetite.